About InovafitoBrasil
The InovafitoBrasil Platform aims to make the production of new phytotherapeutic medicines viable by bringing together various actors within the innovation ecosystem.


The InovafitoBrasil Platform aims to make the production of new phytotherapeutic medicines viable by bringing together various actors within the innovation ecosystem.


The InovafitoBrasil Platform is the result of a broad network of stakeholders, including representatives from the industry, academia, and government.
The initiative stems from the recognition of the latent potential existing in Brazil and the need to create a space for the acknowledgment of national initiatives.


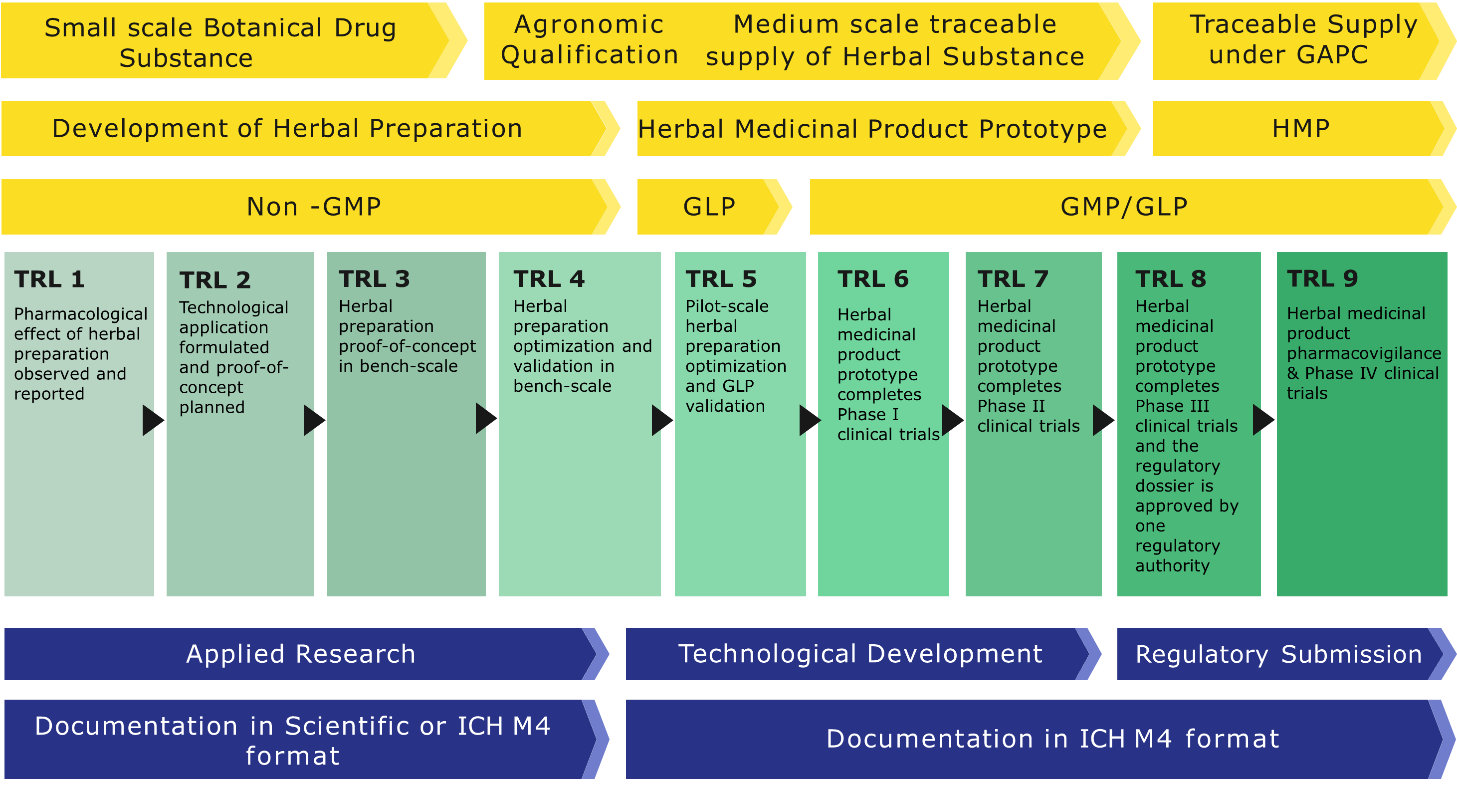
The InovafitoBrasil platform includes a standard roadmap for the development of phytotherapeutic medicaments projects.
The script is based on regulatory requirements and the Technology Readiness Levels (TRLs), which are used by many agencies to determine the need for financial support.
It also has directives for planning and managing projects that generate confidence and reduce investment risks.
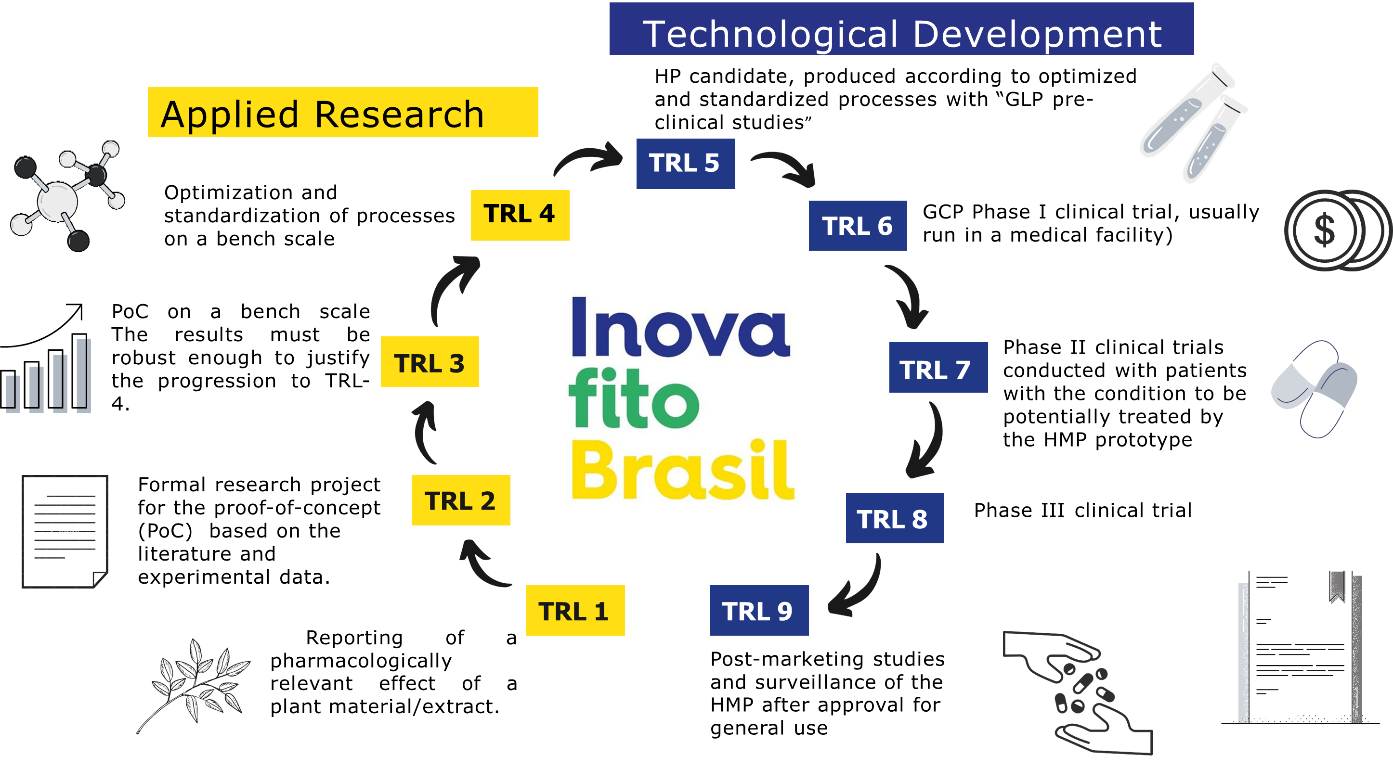
The Technology Readiness Level (TRL) description used in the roadmap for the development projects of phytotherapeutic medicaments registered on the InovafitoBrasil platform is based on field dimensions, active pharmaceutical ingredients (APIs), Good Laboratory Practices (GLP), Good clinical practices (GCP), Good manufacturing practices (GMP), fostering, regulatory compliance, and document formatting.